Bioguide #13
Biofungicides and plant fungal diseases
Using microbial biology to control plant fungal disease.
It’s important to recognise that biofungicides are living organisms with a distinct set of advantages and applications over conventional fungicides. The main advantage of biofungicides is prolonged activity – enabled by the microbe’s ability to colonise the plant/fruit surface and protect the areas where plant fungal disease occurs.
For example, Bacillus-based biofungicides live in the same places and conditions that suit fungal diseases, crowding out the fungus by competing for food and space.
Another advantage of biofungicides is that the Bacillus can access and survive in difficult-to-access areas of the plant. For example, a biofungicide applied early enough, can live inside the grape bunch after it has “closed”, protecting it from botrytis over the long-term when agrichemical sprays cannot reach inside the bunch.
How do biofungicides work
Once applied, biofungicides grow on the plant or fruit surface. When contact is made with a plant fungal disease or bacteria, they begin to produce antimicrobial compounds. In Figure 1, the anti-fungal compounds (produced by Bacillus amyloliquefaciens Bs1b in BioStart’s organic product TripleX) have diffused from the hole in the agar plate and suppressed the growth of the Cladosporium fungus which causes sooty mould. In the case of TripleX, it inhibits Caldosporium spore germination and mycelial growth.
Figure 1: Bioactivity of TripleX against the sooty mould causing fungi Cladosporium.
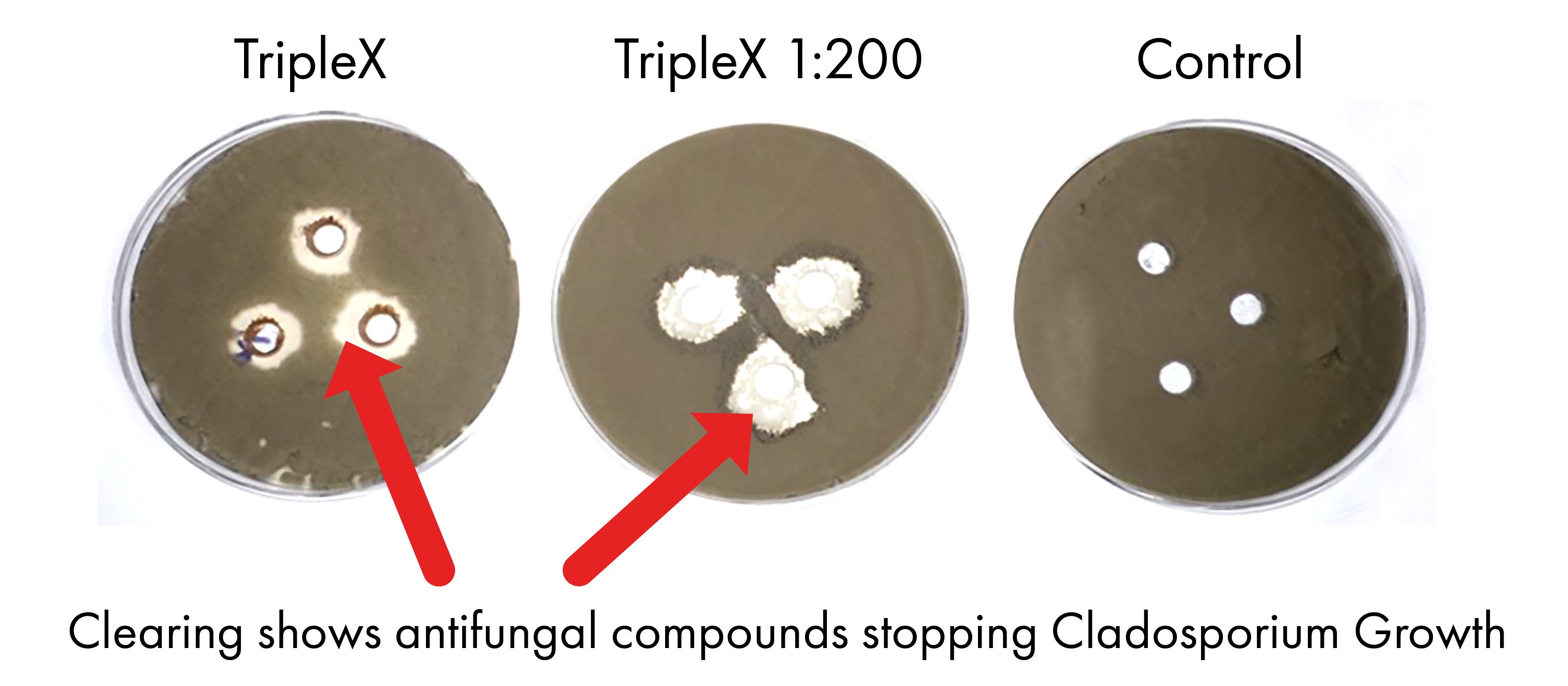
The spores of Cladosporium are spread on to agar plates. Holes are punched into these agar plates and TripleX or water is applied into the holes. The agar plates are incubated for 4 days and fungal growth is observed. Areas of “clearing” around the holes in the agar plates indicate that there were antifungal compounds in those holes that migrated through the agar and inhibited the growth of the plant-fungal disease.
Anti-fungal and anti-bacterial compounds – how biofungicides respond to plant fungal disease
Most biofungicides produce multiple anti-fungal and anti-bacterial compounds that target different biological processes in the disease-causing organism. For example, B. amyloliquefaciens Bs1b in TripleX makes eight different anti-fungal and anti-bacterial compounds (Figure 1), including:
- Cyclic lipopeptides – strong biosurfactants (detergent-like compounds) that disrupt fungal cell walls
- Polyketides – secondary metabolites with broad range anti-microbial activity
- Bacteriocins – small, chemically modified peptides with specific anti-microbial activity.
Figure 2: anti-fungal and anti-bacterial compounds made by Bacillus amyloliquefaciens Bs1b – the active ingredient in BioStart TripleX.
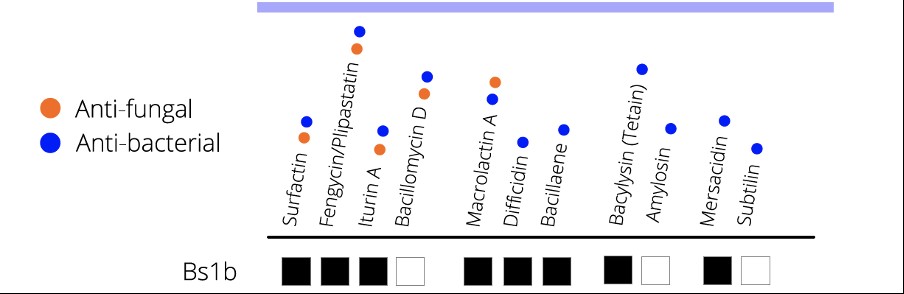
Reduced risk of chemical resistance
A major issue faced by growers is the development of resistance to key agrichemicals by disease-causing organisms. Once this occurs, growers are faced with using multiple different agrichemicals in order to control a single disease. Biofungicides produce multiple anti-microbial compounds (e.g. TripleX makes 8 different compounds), reducing the risk that the plant-fungal disease will develop resistance.
Biofungicides can activate SAR and ISR in the host plant
In addition to directly controlling plant fungal disease, Bacillus biofungicides produce cyclic lipopeptides and polyketides that have been shown to activate the host plant’s Systemic Acquired Resistance (SAR) and Induced Systemic Resistance (ISR) pathways. SAR and ISR are the equivalent of the plant immune system and provide a secondary level of control of plant diseases. Figure 3 shows the compounds produced by the active ingredient in TripleX that activates SAR and/or ISR in plants.
Figure 3: compounds produced by Bacillus amyloliquefaciens Bs1b (the active ingredient in TripleX) that can activate SAR and/or ISR in plants
| Compound | Anti-fungal | Anti-bacterial | Stimulates plant defence mechanisms |
| Surfactin | ++ | ++ | ✓ |
| Plipastatin | +++ | + | ✓ |
| Iturin A | +++ | + | ✓ |
| Macrolactin | + | ++ | ✓ |
Ready to get started?
Check out our range of products dedicated to crop protection:

